Stem Cell Educator Therapy
Introducing: The CB-SC Stem Cell
Stem Cell Educator Therapy would not be possible without CB-SC: human umbilical cord blood-derived multipotent stem cells. Unlike all other stem cells, including hematopoietic, mesenchymal, endothelial progenitor, and monocyte-derived stem cells [4], CB-SC are unique for several distinct reasons. First, they are negative for blood cell lineage markers while expressing embryonic stem cell markers. Second, CB-SC have very low immunogenicity, eliminating the need for genetic stem cell matching [5]. Third, they have the capability to attach tightly to culture dishes with a large, rounded morphology and are resistant to common detaching methods such as trypsin/EDTA. This distinct characteristic allows for a seamless process of collecting suspended lymphocytes treated by CB-SC without the risk of mixing them after ex vivo co-culture [6]. Stem Cell Educator therapy assures every patient that their own “educated” lymphocytes (GleukocellTM) will return to their body during treatment.
Stem Cell Educator Therapy
Dr. Yong Zhao developed Stem Cell Educator Therapy [7] based on his successful results in non-obese diabetic (NOD) mice and other pre-clinical evidence evidence supporting the capability of CB-SC to control autoimmune responses by altering regulatory T cells (Tregs) and human islet beta cell-specific T cell clones isolated from type 1 diabetic patients.
What about safety and efficacy?
Our published and unpublished data [7] has demonstrated Educator Therapy to be entirely safe and non-invasive. Unlike any other stem cell treatment, Educator Therapy does not introduce stem cells or reagents into patients, ensuring 100% immune acceptance and zero risk of rejection.
What Sets Educator Therapy Apart?

- No stem cell injections, transplantations, or blood transfusions occur. Educator Therapy utilizes an in vitro method, ensuring immune acceptance and eliminating the risk of rejection. Stem cells are contained in the Educator Device, ensuring patient’s own (autologous) cells enter the patient’s body.
- CB-SC. Discovered and patented by Dr. Yong Zhao, Educator Therapy is the only technology in the world licensed to use these powerful multipotent stem cells. They are the only stem cells expressed the autoimmune regulator (AIRE) transcription factor with the capability to restore autoimmunity and completely eliminate the requirement for stem cell genetic matching because of their low immunogenicity.
- Zero stem cells come from animals or aborted fetuses. These stem cell treatment methods often result in safety risks as well as ethical and religious concerns. Educator Therapy is ethically responsible.
A Closer Look at Educator Therapy in Type 1 Diabetes
Findings from our clinical trials provide strong evidence that a single treatment of Educator Therapy offers lasting reversal of autoimmunity, allowing the regeneration of islet β cells and improving metabolic control in individuals with long-standing type 1 diabetes [7]. Additionally, individuals experienced markedly improved C-peptide levels, reduced median glycated hemoglobin A1c values (HbA1c), and decreased daily insulin doses. Notably, a single treatment can improve islet β cell function for up to a year.
These trial findings indicate that Educator Therapy with CB-SC meets the scientific expectation that a successful T1D therapy will have the capability to address different arms of the autoimmune response and restore balance to the immune system through systemic and local modulations. This is a capability that Educator Therapy has clearly demonstrated through our clinical data and animal studies [1], [7], [8], [12].
Currently, we received the FDA-approved Regenerative Medicine Advanced Therapy (RMAT) designation and perform clinical studies to improve the efficacy of Educator Therapy in T1D and other autoimmune diseases. For more information, please visit ClinicalTrial.gov.
Case 1: Improvement of β-cell function by Stem Cell Educator therapy. a C-peptide response following a 75-g oral glucose tolerance test (OGTT). A T1D subject (female, 15-year old, 5-year diabetic duration) received 1 treatment with Stem Cell Educator therapy, with one-year follow-up. See following Figure:
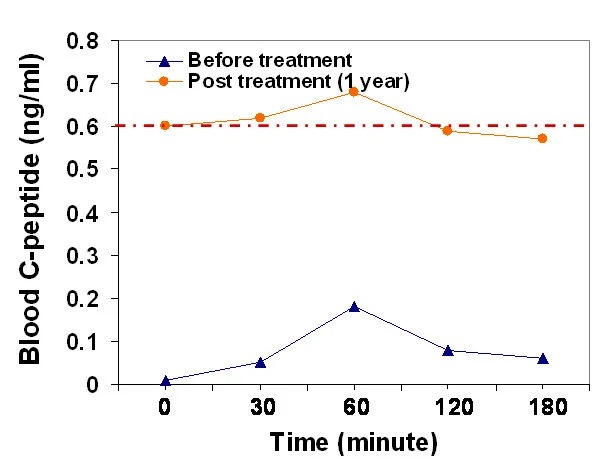
Improve Insulin Sensitivity by Educator Therapy in Type 2 Diabetes
A successful approach to Type 2 Diabetes (T2D) is one that can overcome immune dysfunction and chronic inflammation, as this will be a key tool in addressing the immune response involved in insulin resistance in T2D patients.
In an open-label Phase 1 and Phase 2 study, patients (N = 36) with long-standing T2D were divided into three groups (Group A: oral medications, N = 18; Group B: oral medications + insulin injections, N = 11; Group C: impaired β-cell function with oral medications + insulin injections, N = 7). All patients received one treatment of Educator Therapy.
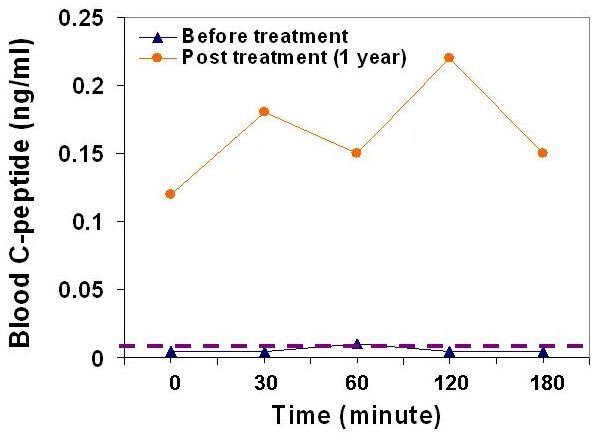
The results of these studies revealed improved metabolic control and reduced inflammation markers. Notably, the homeostasis model assessment of insulin resistance (HOMA-IR) demonstrated improved insulin sensitivity post-treatment (Figure Left). Islet β-cell function in Group C was markedly recovered (Figure Right), and C-peptide levels were restored.
Clinical Case Report—Type 2 Diabetes
Case 1: Before receiving Educator Therapy, patient A (male, 52 years old, 12 years diabetic duration) had been treated with current medications (e.g., metformin, sulfonylureas, pioglitazone) but was unable to control his blood sugar levels (remaining at ~10 mmol/L), despite his insulin dosage increasing to 70 U/day. He had been hospitalized with a minor stroke (left brain). This patient displayed typical insulin resistance, as demonstrated by a 75-g OGTT test with normal levels of fasting and stimulated C-peptide. After receiving Educator Therapy, his insulin dosage decreased by 20 U/day one month later. At 6 months post-treatment, his insulin dosage had continually decreased by 50%, with occurrences of hypoglycemia if he injected a little more insulin. Notably, his HbA1C level was reduced from baseline 9.4% to 7.1% by month 6 (note: 7% is the ADA-recommended standard for diabetics). His fasting blood sugar levels remained at 6–7 mmol/L. This data demonstrates that Educator Therapy can markedly improve insulin sensitivity and overcome insulin resistance (the key issue of T2D).
Case 2: A long-standing diabetic patient B (female, 57 years old, 27 years diabetic duration, 32 U insulin/day) displayed impaired islet beta cell function with a fasting C-peptide level of 0.16 ng/ml (note: 0.6 ng/ml is the lower limit for normal C-peptide levels in Chinese populations). After receiving Educator Therapy for 3 months, her fasting C-peptide level markedly increased to 0.54 ng/ml; correspondingly, her daily insulin dosage decreased by 50%. Her HbA1C level was reduced from baseline 7.3% to 6.4% by month 3. This data indicates that Educator Therapy can restore islet beta cell function and improve metabolic control.

No Stem Cell Injections
Stem Cell Educator Therapy utilizes an ex vivo method, ensuring immune acceptance and eliminating risks of rejection. Stem cells are contained within the Stem Cell Educator Device, ensuring autologous cells enter the patient’s body.
No Stem Cells from Animals or Aborted Fetuses
Many stem cell therapies use stem cells derived from animals or aborted fetuses, which can pose safety risks as well as ethical and religious concerns. Stem Cell Educator Therapy eliminates these issues, thereby opening many markets traditionally closed to stem cell therapies.

Safety
Stem Cell Educator Therapy is a minimally invasive treatment performed outside the patient’s body. Clinical trials demonstrate that this therapy has minimal to negligible side effects.

Proven Research
Over 25 years of research and more than 500 clinical trial participants aged from 3 to 86 years old worldwide demonstrate the safety and efficacy of Stem Cell Educator Therapy.

World’s Only CB-SC Stem Cell Technology
Stem Cell Educator Therapy is the only technology in the world that uses CB-SC, which are more powerful than any other stem cells in their immune-modulating abilities. They can correct autoimmunity, restore immune balance, and eliminate the need for stem cell genetic matching.

Unlimited Expansion of Technology
Stem Cell Educator Therapy has unlimited application potential. Clinical evidence already demonstrates its efficacy for diabetes, alopecia, and other autoimmune diseases. Continued research aims to demonstrate its effectiveness in other rare and incurable diseases.



